11 Mesenteric Disease
Authors: Matt Chia, MD and Nick Mouawad, MD
Mesenteric vascular disease can be broken down into three disease states that we’ll cover today. There’s the arterial disease, which is clearly separated into acute mesenteric ischemia and chronic mesenteric ischemia. Then there is venous disease, which we’ll touch on briefly. There are also a handful of somewhat related diseases that we’ll also sprinkle into these discussions, like median arcuate ligament syndrome and SMA syndrome, but that’s overall where we’re headed.
Mesenteric collatorals are important for protecting against mesenteric ischemia. The gastroduodenal and pacreaticoduodenal arteries connect the celiac and SMA. The meandering artery (Arc of Riolan) and marginal artery of Drummond connect IMA and SMA. Hemorrhoidal branches connect internal iliacs and IMA. (Chandra and Quinones-Baldrich 2010; van Gulik and Schoots 2005)
11.1 Acute Mesenteric Ischemia
11.1.1 Presentation and Evaluation
Can you tell me about the classic presentation and approach to patients presenting with acute mesenteric ischemia?
These patients present with the sudden onset of abdominal pain. Nausea, vomiting, distention, and diarrhea (possibly bloody, described as “sudden and forceful evacuation”) are the common symptoms. Pain out of proportion is the classic buzzword, and can be hidden on multiple choice tests with a pain score of 10/10 with only mild abdominal tenderness on physical exam.
Etiology most often from acute embolism (40-50%), which is often secondary to cardiac source (atrial fibrillation or recent MI).(Wyers, Mark C and Martin 2019)
Vitals typically normal, possible tachycardia.
Lab evaluation typically unremarkable. Leukocytosis, hemoconcentration, and acidosis (high anion gap) all are frequently found, but the absence of these definitely does not rule out acute mesenteric ischemia.
D-dimer has been proposed as a reasonable rule-out test for acute mesenteric ischemia.
How about radiology studies?
Plain film:
Frequently normal, may show ileus in the early stages. In late stage acute mesenteric ischemia, findings on plain film can include bowel wall edema (thumbprinting) or pneumatosis.
CT angio:
Probably represents the most common diagnostic modality to diagnose acute mesenteric ischemia. CTA has the advantages of speed, availability, and noninvasiveness when compared to conventional angiography, and also allows for some assessment of the degree of bowel involvement.
Preferentially, a “negative” oral contrast agent would help prevent oral contrast from causing artifact or obscuring evaluation of the vessels, although the availability of these agents may limit their use.
How about mesenteric duplex (vascular lab studies)?
Mesenteric duplex has the advantage of being able to see the velocity of flow across a stenosis, giving you a good method of quantifying the significance of a stenosis. However, bowel gas often limits the acoustic windows for visualizing the mesenteric arteries, and so we usually will have patients fast for several hours before a study. Also, duplex is more sensitive for proximal disease rather than distal mesenteric involvement. For these reasons, mesenteric duplex is considered the gold standard for evaluating chronic mesenteric ischemia, but has no real role in the evaluation of acute mesenteric ischemia. Can you imagine, having a tech mash a transducer into a patient with acute abdominal pain?!
11.1.2 Management
What’s your approach to the initial management of the patient?
Resuscitation. Fluids, fluids, fluids. These patients are really volume down, and are headed towards a profound distributive shock that will be worsened by your eventual plan for revascularization (think ischemia-reperfusion injury). Also look for electrolyte imbalances and correct those early.
Antibiotics. These patients are not usually septic on initial presentation, but are at high-risk, so broad-spectrum antibiotics with gut coverage (Gram negative and anaerobes) are standard of care.
Heparin. In the absence of a clinical contraindication, these patients should be systemically heparinized, with a bolus, as soon as the diagnosis is made.
Which blood vessel does acute mesenteric ischemia typically involve?
The SMA. This makes sense if you think of each mesenteric distribution. The celiac distribution has organs that have redundant blood supply (like liver and stomach). The IMA is frequently occluded in patients with AAA, but the patients are rarely ever symptomatic due to collateral flow. The hypogastrics (which ARE mesenteric vessels, especially in the situation of an occluded IMA) principally supply the rectum from a mesenteric standpoint.
For embolic pathology, the acute angle of the SMA seems to predispose it to capture emboli from above, but this is more theoretical than proven. The embolism is often lodged distal to middle colic vessel.(Wyers, Mark C and Martin 2019; Kazmers 1998)
Tell me about the two main pathologies, and how they would differ in terms of the anatomy and operative findings.
So the two most common etiologies of acute mesenteric ischemia are embolism and thrombosis.
Embolism is the more common, where preexisting thrombus (think atrial fibrillation, mural thrombus from thoracic aneurysm, etc.) or plaques from atherosclerotic disease break off and lodge in the SMA. The classic operative finding is that an embolism lodges just distal to the middle colic artery, where there is a significant caliber change in the SMA. This is distal to the first few jejunal branches off of the SMA, leading to the classic sparing of the proximal jejunum and transverse colon. In other words, the mid to distal jejunum and all of the ileum will be ischemic, but other areas of the SMA territory are spared. Atherosclerotic debris is typically smaller, and results in smaller, more patchy areas of ischemia.
Thrombosis occurs primarily as a plaque rupture of preexisting atherosclerotic disease, resulting in acute thrombosis at the site of the disease. Thus the patients will often present with an acute-on-chronic symptomatology, having classic symptoms of postprandial abdominal pain, food fear, and weight loss, but with a sudden onset of severe symptoms. This frequently allows for the development of mesenteric collaterals, which may make the onset of acute symptoms more insidious than for embolic pathology. In the majority of situations, the atherosclerotic disease is most severe right at the origin (consistent with what we know about shear stress and branch points in blood vessels). This means that when the plaque ruptures, the entirety of the SMA occludes, leading to ischemia of the entire territory, as opposed to the jejunal-sparing distribution seen in embolic disease.
Describe the operative steps to getting exposure of the supraceliac aorta (or the celiac artery).
Divide triangular ligament to mobilize left lobe of liver
Divide gastrohepatic ligament to enter the lesser sac
Retract liver to right with a self-retaining retractor
Push esophagus left (use NGT to assist with identification)
Divide peritoneum overlying crura to identify celiac vessels
Typically trace common hepatic artery backwards to identify celiac artery
Watch out for the left gastric vein as it crosses the celiac artery as it drains the lesser curve of the stomach into the portal vein.
About half of the time, the phrenic artery takes an origin from the celiac artery and must be controlled during exposure.
To expose the supraceliac aorta, divide the median arcuate ligament and separate the left and right crura from each other.
So through this kind of exposure, what mesenteric vessels do you get access to?
You can trace most of the proximal celiac distribution right at the origin, and through this exposure you get access to the origin of the SMA if you mobilize the superior border of the pancreas. The neck of the pancreas and the splenic vein cross the anterior of the SMA, obscuring the rest of the mid and distal SMA from the superior approach.
How about getting to the rest of the SMA?
There are a couple of places you can get exposure to the SMA.
Most commonly you’ll hear it described at “root of the mesentery.” Specifically, lifting up the transverse colon will stretch out its mesentery (i.e. transverse mesocolon). At the bottom, or “root” of the transverse mesocolon, a transverse incision is made. If the middle colic artery is palpable in the mesocolon, the incision can be made around it, and you can trace the middle colic backwards to the SMA. Usually you’ll find the SMV first, and the SMA will be just to the left of it. Be sure to identify and preserve small jejunal branches during the dissection. If needed, careful dissection superiorly, going behind the inferior border of the pancreas can get a little more proximal exposure. Embolectomy through a transverse arteriotomy in the SMA is the best approach.(Wyers and Martin 2019; Kazmers 1998)
Alternatively, you can get to the SMA from a lateral approach, specifically from the left side. Begin by dividing the ligament of Treitz and mobilizing the 4th portion of the duodenum. The SMA is found in the tissues just cephalad to the duodenum. You can also improve your proximal exposure if needed by retracting the inferior border of the pancreas cephalad to the level of the left renal vein.
Other options include a retroperitoneal exposure, like you were preparing to treat a thoracoabdominal aneurysm. Additionally, the more distal SMA can just be identified in the small bowel mesentery.
So for the operative strategy for acute mesenteric ischemia, tell me about the general approach to the patient.
Resect frankly necrotic bowel and contain gross spillage. Once you revascularize the bowel, compromised-appearing bowel may improve and not need immediate resection. Thus the first step is only damage control, to remove anything completely unsalvageable that is making the patient sick, or anything causing gross contamination of the operative field. The key is you’re not doing anything definitive with the bowel as your first step.
Revascularization. SMA embolectomy is the initial management of choice for embolic disease. Thrombotic disease, on the other hand, may be more challenging to treat by embolectomy alone, and frequently are treated with a bypass. (More to come on these procedures).
Re-assess bowel viability. Clinical status permitting, 20-30 minutes should be taken to fully assess the results of the revascularization before proceeding with resection. Perfusion can be assessed by many methods, including clinically, by Doppler, pulse oximetry (a.k.a. photoplethysmography), fluorescein fluorescence, etc. The take home is to give the bowel enough time to be perfused before going ahead with resection.
Proceed with temporizing or definitive bowel repair. Resection, leaving in discontinuity, primary anastomosis, diversion, etc. All of these are options on the table, but the key here is that all of the previous steps occur before addressing the bowel.
Consider second-look laparotomy. Many times, bowel may look questionable even after revascularization and thorough re-assessment. To preserve the most bowel length, it may be reasonable to leave borderline bowel alone at the index operation and do a “second look” to fully reassess the bowel, especially after the patient has benefited from aggressive resuscitation in the ICU.
Endovascular approaches to acute mesenteric ischemia have been described but data is limited. Exploratory laparotomy and embolectomy should be preferred management.(Wyers and Martin 2019)
Let’s talk a little about the steps for an SMA embolectomy.
Typically, you’ll expose at the root of the transverse mesocolon. After obtaining proximal and distal control, an arteriotomy is made, and embolectomy can be performed by passing Fogarty catheters in both a retrograde and antegrade fashion. The arteriotomy can be made transversely for an embolectomy, and thus could be closed primarily. A longitudinal arteriotomy may be advisable if you have a high suspicion that you’ll need to do a bypass, and if not, may be closed with vein patch angioplasty (remember that the field is contaminated or dirty in many situations).
So if I’m gonna do a bypass, what are my options for conduit?
Yeah, so again, because the field is frequently contaminated or dirty, a good conduit is saphenous, followed by femoral vein. Thus every patient undergoing surgery for acute mesenteric ischemia should have both legs prepped out in the field. Prosthetic conduit has the advantages of being more resistant to kinking (externally reinforced), likely better patency than vein (although data are a little mixed), but in the situation of gross contamination may be less preferred than vein. Other less common options include cryo-preserved cadaveric homograft, or rifampin-soaked prosthetic.
And what are some of my options for constructing a bypass?
Short retrograde aorto-SMA bypass:
This bypass takes is origin off of the aorta just below the SMA, anastomosing typically end-side onto the SMA just below its origin in order to bypass ostial or very proximal disease. This is a relatively quick bypass, with only one field of dissection directly from the aorta below the SMA onto the proximal-mid SMA. The length of the bypass is very short, limiting concerns with kinking or twisting of the bypass. However, this may not always be feasible, as SMA disease often coexists with significant aortic disease. Additionally, the other bypasses described have better reported patency.(Scali et al. 2019)
Long retrograde R iliac-SMA bypass (“C-loop”):
This bypass originates from the right common iliac artery, which presents a number of distinct advantages over an aorto-mesenteric bypass. First, using the iliacs avoids the hemodynamic consequences of an aortic cross-clamp, which may be contraindicated depending on your patient’s medical condition. Second, you can avoid showering, causing dissection, or otherwise injuring your clamp sites if you have significant disease in the mesenteric segment of the aorta, which is common in patients with chronic mesenteric ischemia. The graft should be tunneled in a gentle C-loop towards the SMA to avoid kinking or twisting. The proximal anastomosis is performed end-side on the iliac artery, and the distal can be performed either end-end or end-side depending on the anatomy of the disease. Especially when using prosthetic in a contaminated field, you can consider taking an omental flap to wrap or cover the prosthetic.
Antegrade supraceliac aorta-SMA bypass:
This bypass originates from the supraceliac aorta. If revascularization of both celiac and SMA is planned, a bifurcated graft can be selected. A side-biting aortic clamp can be used to mitigate the hemodynamic effect of an aortic cross-clamp. The tunnel to the SMA is created with gentle finger dissection in a retropancreatic plane, taking care to avoid injury to the SMV.
What other options have been described for treatment of acute mesenteric ischemia?
Retrograde open mesenteric stenting (ROMS)
So ROMS is a hybrid procedure involving an upper mid-line laparotomy that is used to evaluate the bowel. Through this incision, SMA exposure is obtained just as in a traditional open fashion at the root of the mesentery. The mid-SMA is then punctured under direct vision, and the area of disease is attempted to be treated from a retrograde approach back into the aorta. If bowel ischemia is found, the upper mid-line is easily lengthened into a traditional vertical laparotomy incision.(Milner, Woo, and Carpenter 2004; Blauw et al. 2014; Gustavo S. Oderich et al. 2018)
Endovascular treatment (percutaneous thrombectomy / thrombolysis / pharmacomechanical thrombectomy)
Some authors have described completely endovascular approaches to treatment of acute mesenteric ischemia. However, the major limitation is the inability to assess the bowel. These patients are frequently those who are deemed to be lower-risk for frank bowel ischemia or perforation, but the rates of laparotomy and bowel resection after these treatments have been described very high rates. Probably not your first answer for oral boards.(Lim et al. 2019)
11.1.3 Other Etiologies
What are some other, more rare etiologies of acute mesenteric ischemia?
Embolism is the most common etiology of acute mesenteric ischemia (40-50%), and thrombotic etiology composes another quarter to a third of these populations. The other two etiologies to consider are non-occlusive mesenteric ischemia and mesenteric venous thrombosis.
Perfect, so how does a non-occlusive mesenteric ischemia (or NOMI) patient differ from what we’ve been talking about?
So NOMI patients typically do not have a focal lesion like you see with embolism or thrombosis. What happens to these patients is that classically they’re pretty sick patients with some predisposing factors, most commonly ESRD. On top of that, there was some clear inciting hemodynamic event causing sustained hypotension, such as recently getting a session of hemodialysis or undergoing cardiopulmonary bypass. The presentation is more indolent and less obvious than embolic or thrombotic acute mesenteric ischemia, and the imaging findings are more consistent with a diffuse vasospasm and hypovolemia picture.
Treatment is primarily conservative, with the emphasis on resuscitation and addressing whatever the underlying etiology is. Adjuncts to this include placement of infusion catheters into the affected vessel with infusions of vasodilators (most commonly papaverine at 30-60mg/hr) or prostaglandin (Wyers, Mark C and Martin 2019; Trompeter et al. 2002) NOMI with portal venous gas, pneumatosis and free air often indicate need for laparotomy.
How about mesenteric venous thrombosis?
This is the most rare and most difficult to diagnose. They have a very slow course, frequently with a lot of other workup already done. There’s a wide variety of causes that have been reported, and any of the things that contribute to Virchow’s triad have been reported (thrombophilia from coagulopathy or malignancy, venous stasis from abdominal hypertension or obesity, direct injury from trauma, surgery, or inflammation). The diagnostic test of choice is a CT with portal vein contrast, which most commonly identifies thrombosis in the superior mesenteric vein (but can also involve the IMV, portal vein, or splenic vein). The treatment of choice is therapeutic anticoagulation.(Acosta and Björck 2019)
11.2 Chronic Mesenteric Ischemia
11.2.1 Presentation and Evaluation
How does chronic mesenteric ischemia differ in its presentation?
Chronic mesenteric ischemia is characterized by post-prandial abdominal pain, typically 30-60 minutes after eating (think after gastric emptying time). This pain is usually severe, crampy, and resolves after minutes to hours of time. The pain leads to food fear and eventual unintentional weight loss. The next step in evaluation in these patients is a mesenteric duplex to evaluate for elevated velocities.
However, the clinical presentation here is key. Chronic mesenteric ischemia is pretty unlikely in patients who do not have this constellation of clinical symptoms, and it’s really common for patients to be referred with imaging findings of elevated mesenteric velocities on duplex who have none of the clinical findings, and thus do not benefit from any intervention.
What are the duplex criteria for chronic mesenteric ischemia?
Yeah, so really the two vessels we’re concerned with here are the celiac artery and the SMA. It’s pretty rare that stenosis of the IMA results in clinically significant ischemia, and also pretty rare that isolated disease of either the celiac or the SMA would be enough to cause significant symptoms. Typically you see the classic symptomatology of chronic mesenteric ischemia in patients with occlusive disease in both the celiac and the SMA.
The key numbers to remember here are peak systolic velocities (that’s PSV) of 200 cm/s for the celiac artery, and 275 cm/s for the SMA. These correspond to a stenosis of at least 70%. Remember, the superior mesenteric artery has higher velocity criteria, in case you forget. These classic numbers come from a study done in 1993, and many other studies have demonstrated other thresholds. Make sure to check with your friendly neighborhood vascular technician to see what the thresholds in your local lab are!
In addition to the PSV, you can also use the end diastolic velocity (EDV) to predict stenosis. For the EDV values in the celiac and the SMA, think approximately 50 cm/s predicts a stenosis of 50% (easy to remember, 50-50). This is known as Bowersox criteria, and is actually >45, but 50 is easier to remember.(Bowersox et al. 1991)
You can also predict a higher degree of stenosis with EDV values. This is easy for the SMA, where an EDV of 70 cm/s predicts a stenosis of 70% (70-70). For the celiac, it’s a bit higher, where an EDV of 100 cm/s predicts that same 70% stenosis.
So to review the numbers: (Moneta et al. 1991, 1993; Zwolak et al. 1998; AbuRahma et al. 2012)
PSVs: 200 in the celiac or 275 in the SMA gives you 70% stenosis
EDVs: 50 in the celiac or 50 in the SMA gives you 50% stenosis
EDV #2: 100 in the celiac, or 70 in the SMA gives you 70% stenosis.
Final thought. When the celiac artery is severely narrowed or occluded, often you’ll have retrograde flow coming from the SMA via collaterals (these are your pancreaticoduodenal arcades via the GDA). You can see this as backwards flow in the common hepatic artery, and this has 100% specificity for disease in the celiac artery.(Warncke et al. 2019)
11.2.2 Management
What’s next in the approach to these patients?
Frequently they’re going to get some kind of axial imaging, either CTA or MRA, to help plan for their revascularization, although this isn’t strictly necessary. Often endovascular approaches are the mainstay of treatment. Remember that the angle of the SMA frequently favors a brachial approach, although you can also engage the SMA from a femoral approach with preformed catheters (think Sos, Cobra, etc) or steerable sheaths. Remember to watch out for median nerve compression from brachial sheath hematomas if you’ve opted for a percutaneous brachial approach, with a low threshold to evacuate the hematoma and perform median nerve decompression if the patient develops and symptoms in the median nerve distribution postoperatively. Also remember that a brachial sheath hematoma will not be easily palpable or visible at the skin level, so the neurologic exam postoperatively is key.
Single vessel SMA revascularization is widely accepted for chronic mesenteric ischemia without necessarily needing to revascularize the celiac axis. (Gustavo S. Oderich and Ribeiro 2019; Foley et al. 2000)
Any other technical tips for endo intervention on the mesenteric arteries?
The best view for diagnostic angiography in mesenteric ischemia is the lateral view.
Most often balloon-expandable stents are selected for the mesenteric circulation. The precision of deployment and the increased hoop strength of a balloon expandable stent make them more favorable here over the radial force of a self-expanding stent. You can also flare the proximal end of a balloon-expandable stent into the aorta, making sure that you’ve really treated any ostial disease. Some authors advocate for use of embolic protection devices to prevent distal embolism, and other authors also advocate for the use of covered stents for the mesenteric circulation due to better patency.(Gustavo S. Oderich et al. 2013; Gustavo S. Oderich and Ribeiro 2019)
What is the surveillance protocol for an SMA stent?
At most institutions it is 1mo, 6mo and annually thereafter.(Mohler et al. 2012; Zierler et al. 2018)
How about open revascularization?
In most centers, up to 80% of CMI is treated endovascularly. Open is often reserved for those that fail endovascular therapy.(Gustavo S. Oderich, Gloviczki, and Bower 2010; Gustavo S. Oderich and Ribeiro 2019)
So we talked a lot about open revascularization earlier in the acute mesenteric ischemia segment. The techniques are broadly similar, with the exception that often for chronic mesenteric ischemia, frequently you’ll be planning to revascularize both the celiac and the SMA. The approach for this is with a bifurcated graft from the supraceliac aorta, taking one limb down to the celiac artery, and the other limb tunneled in a retropancreatic fashion to the mid-SMA. Remember that this approach requires a supraceliac aortic cross clamp, which your patient may not be able to tolerate. But single revascularization of SMA is often acceptable with appropriate collaterals. Taking a retrograde bypass off of the common iliac artery as we previously described may be a better option for patients with significant comorbidities. A further option that completely avoids a bypass is to go right for a trapdoor endarterectomy of the celiac artery and the SMA, allowing you to address coral reef or proximal/ostial disease (such as flush occlusions) that are difficult to treat endovascularly.
11.3 Visceral Aneurysms
Aneurysms can occur in a diverse range of vascular beds in the visceral vessels. Size criteria for repair is dependent on the location and the etiology, with some aneurysms mandating repair at any size (gastric, gastroepiploic, gastroduodenal, pancreaticoduodenal, SMA, colic), and others considered appropriate to observe in asymptomatic patients until certain size thresholds are met. The SVS clinical practice guidelines are the optimal resource to review these criteria.(Chaer et al. 2020)
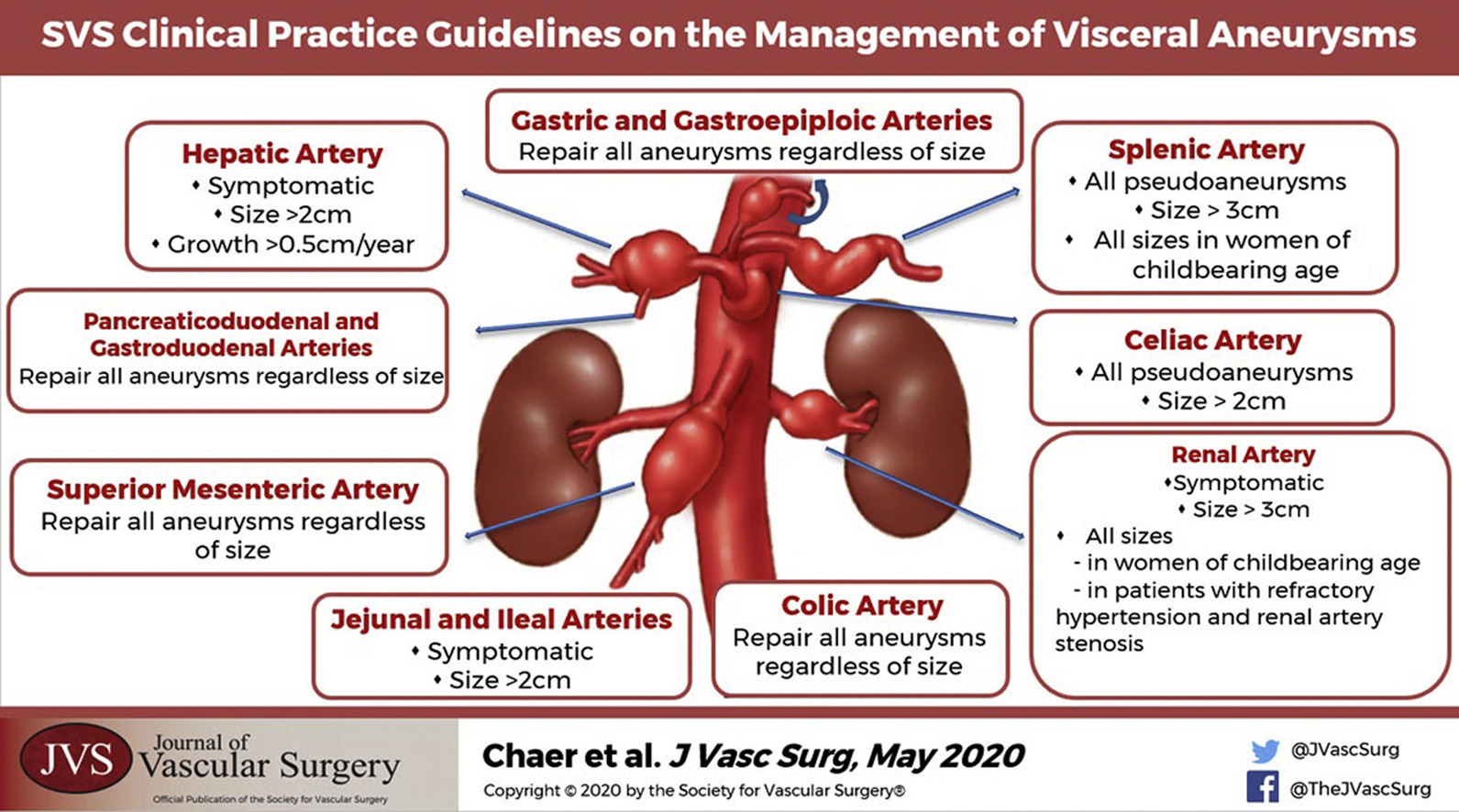
11.3.1 SMA Aneurysm
If symptomatic, most SMA aneurysms are mycotic in nature and require treatment. Treat with resection and saphenous vein interposition and 6 weeks antibiotic therapy based on intraoperative cultures.(Lee et al. 2008 Nov-Dec)
11.3.2 Splenic Aneurysm
Repair all pseudoaneurysms, or if size > 3cm, or at any size if of childbearing age. Consider splenectomy vaccines pre-treatment (covers encapsulated organisms Shigella, Haemophilus influenzae, and Neisseria).
Post splenic aneurysm embolization, L flank pain in stable patients likely represents splenic infarction. Treat with hydration and analgesia. Rates are lower in stented patients.(Kwong, Rockman, and Kashyap, n.d.; Piffaretti et al. 2007; Zhu et al. 2019)
Splenic artery embolization also an effective way to treat thrombocytopenia, particularly chemo induced, when splenectomy is too high risk.(Bhatia et al. 2015)
11.4 Median Arcuate Ligament Syndrome (MALS)
Can you tell me about median arcuate ligament syndrome and how that differs from chronic mesenteric ischemia?
Median arcuate ligament syndrome (MALS), which has many names (Dunbar syndrome, celiac axis compression syndrome, etc.) is a somewhat controversial entity that occurs when repeated compression of the celiac artery occurs against the median arcuate ligament during respiratory variation. The thing you want to visualize here is that during full exhalation, the lungs are completely emptied, and the diaphragm moves up at a sharper angle. This angulation kinks off the celiac artery more severely, so velocities in exhalation are higher in MALS. However, these findings are common in asymptomatic patients, and so just like chronic mesenteric ischemia, the clinical presentation is key. They’ll have a similar presentation as chronic mesenteric ischemia patients, with post-prandial pain, food fear, and weight loss, but often the symptomatology is a little more indolent in these patients. Because of this, these patients have frequently gotten the million dollar workup for nonspecific GI pain. Duplex ultrasound of shows celiac PSV >200 and EDV >50, normalizes during deep inspiration, and post-stenotic spectral broadening.(Zwolak et al. 1998)
The treatment of choice for suspected MALS is a laparoscopic median arcuate ligament release, frequently performed by a MIS/foregut surgeon. A key point here is that you don’t want to be fooled into putting a stent in these patients before they’ve gotten their median arcuate ligament release, because the dynamic motion of the diaphragm is likely to crimp or bend the stent if that hasn’t been treated yet. It may be that some patients benefit from endovascular treatment after release, though, as some think that the chronic damage from MALS can result in intimal damage/scarring that persists even after the extrinsic compression is treated by the median arcuate ligament release.
11.5 SMA Syndrome
What is SMA syndrome?
SMA syndrome, also called Wilkie’s Syndrome is a rare entity where the 3rd portion of the duodenum gets compressed between the SMA and the aorta, causing a functional gastric outlet obstruction. Patients are typically emaciated, having lost a significant amount of weight before their symptom onset. What’s happened is they’ve lost the retroperitoneal fat pat that normally surrounds the SMA, and so the angle between the SMA and the aorta becomes more acute, pinching off the duodenum.
Classic findings in fluoroscopic swallow in SMA syndrome are delayed gastric emptying, a dilated duodenum, to and fro peristalsis in the proximal duodenum and cutoff in the third portion. EGD can show pulsation in the 3rd portion of the duodenum.(Warncke et al. 2019)
The treatment of choice here is enteral feeding with a nasojejunal tube or other surgically placed tube and TPN, because what really will help them here is weight gain. GJ bypass or duodenal mobilization is sometimes needed for treatment, but is controversial. It’s not really a mesenteric vascular disease, but it sometimes shows up on exams as a related entity.(Welsch, Büchler, and Kienle 2007; Merrett et al. 2009)
11.6 Uterine Fibroids
Symptomatic uterine fibroids are now often treated with uterine artery embolization. Many patients require bilateral uterine artery embolization (UAE), but may only need unilateral if blood supply is unilateral. Infarcted tissue can be expelled or become infected even years after initial embolization. (Stępniak 2018)